Ionic EquilibriumHard
Question
The self ionisation constant for pure formic acid, K = [HCOOH2+][HCOO-] has been estimated as 10-6 at room temperature. The density of formic acid is 1.15 g/cm3.The percentage of formic acid molecules in pure fomic acid are converted to formate ion:
Options
A.0.002%
B.0.004%
C.0.006%
D.0.008%
Solution
Given density of formic acid = 1.15 g/cm3
∴ Weight of formic acid in 1 litre solution = 1.15 × 103 g
Thus, [HCOOH] =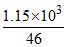 = 25 M
= 25 M
Since in case of auto ionisation
[HCOOH2+] = [HCOO-] and [HCOO-] [HCOOH2+] = 10-6 ⇒ [HCOO-] = 10-3
Now % dissociation of HCOOH =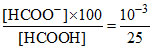 × 100 = 0.004%.
× 100 = 0.004%.
∴ Weight of formic acid in 1 litre solution = 1.15 × 103 g
Thus, [HCOOH] =
Since in case of auto ionisation
[HCOOH2+] = [HCOO-] and [HCOO-] [HCOOH2+] = 10-6 ⇒ [HCOO-] = 10-3
Now % dissociation of HCOOH =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A aqueous solution contains an unknown concentration of Ba2+. When 50 mL of a 1 M solution of Na2SO4 is added, BaSO4 jus...A 2.5 g impure sample containing weak monoacidic base (Molecular weight = 45) is dissolved in 100 ml water and titrated ...Calculate [S2–] in a solution originally having 0.1 M – HCl and 0.2 M – H2S. For H2S, Ka1 = 1.4 × 10−7 and Ka2 = 1.0 × 1...After solid SrCO3 was equilibrated with a buffer at pH 8.6, the solution was found to have [Sr2+] = 2.0 × 10−4 M, what i...For the gas phase reaction $\text{C}_2\text{H}_4 + \text{H}_2 \rightleftharpoons \text{C}_2\text{H}_6$ ($\Delta H = -32....