Ionic EquilibriumHard
Question
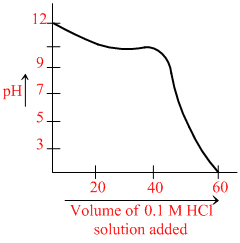
When weak base solution (50 mL of 0.1 N NH4OH) is titrated with strong acid (0.1 N HCl), the pH of the solution initially decreases fast and then decreases slowly till near the equivalence point (as shown in figure). Which of the following is/are correct.

Options
A.The slow decrease of pH is due to the formation of an acidic buffer solution after the addition of some HCl.
B.The slope of shown graph will be minimum when 25 mL of 0.1 N HCl is added.
C.The slow decrease of pH is due to the formation of basic buffer solution.
D.The initial fast decrease in pH is due to fast consumption of OH- ions by HCl.
Solution
(a) is wrong.
For reaction of SA with WB, basic is formed β is maximum. When pOH = pKb.
At 50% neutralisation (25 mL of 0.1 N HCl)
Slope of the given graph will be least and the buffer will have maximum buffer capacity
For reaction of SA with WB, basic is formed β is maximum. When pOH = pKb.
At 50% neutralisation (25 mL of 0.1 N HCl)
Slope of the given graph will be least and the buffer will have maximum buffer capacity
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
At infinite dilution the percentage dissociation of both weak acid and weak base is:...Which one of the following statements is not true?...The base imidazole has a Kb of 1.0 × 10–7 at 25°C. In what volumes should 0.02 M-HCl and 0.02 M imidazole be mixed to ma...1.4 g of an organic compound was digested according to Kjeldahl′s method and the ammonia evolved was absorbed in 6...A certain weak acid has a dissociation constant of 1.0 × 10-4. The equilibrium constant for its reaction with a str...