Coordination CompoundHard
Question
Which of the following complex does not show geometrical isomerism ?
Options
A.[Co (NH3)4 Cl2]+
B.[Co (NH3)3 (NO2)3]
C.[Cr (en)3]3+
D.[Pt (gly)2]
Solution
(A) There are two geometrical isomers, cis and trans.
(B) There are two geometrical isomers, facial and meridional.
(C) ′en′ is a symmetrical ligand, and M(AA)3 has only one form.
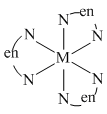
(D) There are two geometrical isomers because glycine is an unsymmetrical ligand.
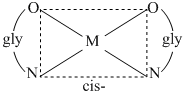
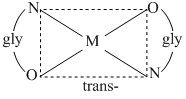
(B) There are two geometrical isomers, facial and meridional.
(C) ′en′ is a symmetrical ligand, and M(AA)3 has only one form.

(D) There are two geometrical isomers because glycine is an unsymmetrical ligand.


Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The formula for the compound tris (ethane-1, 2-diamine)cobalt (III) sulphate is...The oxidation sate of iron in Na4[Fe(CN)5(NOS)] is...The complex ion which has no d electrons in the central metal atom is (At No. Cr = 24, Mn = 25, Fe = 26 , Co = 27) :...Which of the following is correct for the complex [NiBr2(PPh3)2] ?...Which of the following complex species is not expected to exhibit optical isomerism ?...