Gaseous StateHard
Question
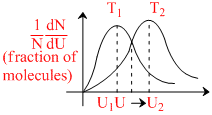
In the above maxwellian plot at two different temperature which of the following statements may be true

Options
A.Area under the two plots is the same
B.Fraction of molecules with speed u1 at T1 > fraction of molecules having speed u2 at T2
C.U2 > U1 and T2 > T1
D.UMPS at T1 < UMPS at T2 ; URMS at T1 < URMS at T2
Solution
With increase in temperature, most probable velocity increases & fraction of molecules with velocity equal to M.P. velocity decreases. Total no. of molecules remain same.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
I, II, III are three isotherms respectively at T1, T2 and T3 as shown in graph. Temperature will be in order...Kinetic theory of gases proves...Coordination number of Zn2+ and S2- in the crystal structure of wurtzite are...The inversion temperature of hydrogen is (given van der Waal′s constants a and b are 0.244 atmL2mol-2 and 1.027 L ...50 mL of each gas A and of gas B takes 200 and 150 s respectively for effusing through a pin hole under the similar cond...