Gaseous StateHard
Question
LPG is a mixture of n - butane & iso - butane. The volume of oxygen needed to burn 1 kg of LPG at NTP would be :
Options
A.2240 l t
B.2510 l t
C.1000 l t
D.500 l t
Solution
C4H10 + 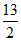 O2 → 4 CO2 + 5 H2O
O2 → 4 CO2 + 5 H2O
x ml n-butane
y ml isobutane
Volume of O2 = x ×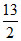 + y ×
+ y × 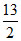
x ml n-butane
y ml isobutane
Volume of O2 = x ×
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Which one of the following statements is NOT true about the effect of an increase in temperature on the distribution of ...A bottle of dry ammonia and a bottle of dry hydrogen chloride connected through a long tube are opened simultaneously at...A sample of H2 collected over H2O at 27oC and a pressure of 591 mm Hg has a volume of 240ml. what volume would the dry H...For one mole of a van der Waals gas when b = 0 and T = 300 K, the PV vs. 1/V plot is shown below. The value of the van d...For the reaction at 300 K at 1 bar2A(g) + B (g) → 2C(g)ᐃHof [A(g)] = -110 calᐃHof [B(g)] = - 50 cal...