Chemical BondingHard
Question
Which of the following species does not exist under normal condition ?
Options
A.Be2+
B.Be2
C.B2
D.Li2
Solution
Molecules with zero bond order, do not exist.
(a) Be2+(4 + 4 - 1 = 7)
= σls2, σ*ls2, σ2s2, σ*2s1
BO =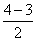 = 0.5
= 0.5
(b) Be2 (4 + 4 = 8 )
= σls2, σ*ls2, σ2s2, σ*2s2
BO =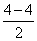 = 0
= 0
(c) B2 (5 + 5 = 10)
= σls2, σ*ls2, σ2s2, σ*2s2, π2px1 π2py1
π2py1
BP =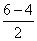 = 1
= 1
(d) Li2 (5 + 5 = 10)
= σls2, σ*ls2, σ2s2
BO =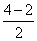 = 1
= 1
Thus, Be2 does not exist under normal condition.
(a) Be2+(4 + 4 - 1 = 7)
= σls2, σ*ls2, σ2s2, σ*2s1
BO =
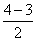 = 0.5
= 0.5(b) Be2 (4 + 4 = 8 )
= σls2, σ*ls2, σ2s2, σ*2s2
BO =
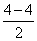 = 0
= 0(c) B2 (5 + 5 = 10)
= σls2, σ*ls2, σ2s2, σ*2s2, π2px1
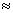 π2py1
π2py1BP =
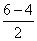 = 1
= 1(d) Li2 (5 + 5 = 10)
= σls2, σ*ls2, σ2s2
BO =
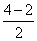 = 1
= 1Thus, Be2 does not exist under normal condition.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of the following oxides is amphoteric in character?...Select the correct statements -...Assuming that Hund′s rule is violated, the bond order and magnetic nature of the diatomic molecule B2 is...Pick out the correct statement with respect to [Mn(CN)6]3 –:-...Which of the following hydrogen bonds is the strongest?...