NEET | 2017Chemical BondingHard
Question
Pick out the correct statement with respect to [Mn(CN)6]3 –:-
Options
A.
It is sp3d2 hybridised and tetrahedral
B.
It is d2sp3 hybridised and octahedral
C.
It is dsp2 hybridised and square planar
D.
It is sp3d2 hybridised and octahedral
Solution
[Mn(CN)6]3- O.S. of Mn is (+3)
Mn+3 3d4 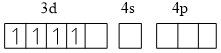
Presence of SFL (Pairing is possible)
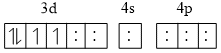 [d2sp3, octahedral]
[d2sp3, octahedral]
t2g eg
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Match the column...The graph between log(x/m) and log P is a straight line at angle 45o with intercept OA as shown. Hence (x/m) at a pressu...Hg(NO3)2 W + X + O2X + H2O → NHO2 HNO3...A reacts according to the following n Ist order reaction simultaneously :- Then is :-...Erich of the following molecule has highest bond energy ?...