Gaseous StateHard
Question
Two glass bulbs A (of 100 mL capacity), and B (of 150 mL capacity) containing same gas are connected by a small tube of negligible volume. At particular temperature the pressure in A was found to be 20 times more than that in bulb B. The stopcock is opened without changing the temperature. The pressure in A will
Options
A.drop by 75%
B.drop by 57%
C.drop by 25%
D.will remain same
Solution
nT = nA + nB
PfVf = PAVA + PBVB
Pf =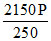
Decrease in P of A = 20 P -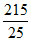 P
P
% drop =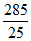 P ×
P × 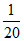 × 100 = 57%.
× 100 = 57%.
PfVf = PAVA + PBVB
Pf =
Decrease in P of A = 20 P -
% drop =
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Coordination number of Zn2+ and S2- in the crystal structure of wurtzite are...The R.M.S. speed of the molecules of a gas of density 4 kg m-3 and pressure 1.2 ×105 N m- 2 is :...Which one of the following statements is NOT true about the effect of an increase in temperature on the distribution of ...Dominance of storage repulsive forces among the molecules of the ges (Z = compressibility factor):...At high pressure, the compressibility factor (Z) is equal to :-...