Gaseous StateHard
Question
For two gases, A and B with molecular weights MA and MB, it is observed that at a certain temperature, T, the mean velocity of A is equal to the root mean square velocity of B. Thus the mean velocity of A can be made equal to the mean velocity of B, if
Options
A.A is at temperature, T1 and B at T2 T1 > T2
B.A is lowered to a temperature T2 < T while B is at T
C.Both A and B are raised to a higher temperature
D.Both A and B are lowered in temperature
Solution
Given 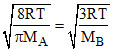 ⇒ 8MB = 3π MA
⇒ 8MB = 3π MA
&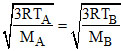 ⇒
⇒ 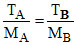 ⇒ MB . TA = MA . TB
⇒ MB . TA = MA . TB
⇒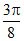 MA . TA = MA . TB ⇒ TB > TA Hence (B)
MA . TA = MA . TB ⇒ TB > TA Hence (B)
&
⇒
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
NH3 gas is liquefied more easily than N2. Hence:...A gas cylinder containing cooling gas can withstand a pressure of 14.9 atmosphere. The pressure gauge of cylinder indica...Consider the following statements: If the van der Waal′s parameters of two gases are given as a (atm lit2 mol-2) b...Dominance of storage repulsive forces among the molecules of the ges (Z = compressibility factor):...A certain volume of argon gas (Mol. Wt. = 40) requires 45 s to effuse through a hole at a certain pressure and temperatu...