Chemical Kinetics and Nuclear ChemistryHard
Question
For an endothermic reaction where áƒH represents the enthalpy of the reaction in kJ/ mol, the minimum value for the energy of activation will be :
Options
A.less than áƒH
B.zero
C.more than áƒH
D.equal to áƒH
Solution
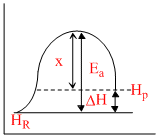
For an endothermic reaction
Ea = áƒH + x
x may be taken as energy released in the formation of products from transition state and thus Ea will be always greater than áƒH in the endothermic reaction it eill be always less than Ea .
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
If I is the intensity of absorbed light and C is the concentration of AB for photochemical process. AB + hv → AB*,...The correct order of thermal stability of hydroxides is :...In the reaction N2 + 3H2 ⇋ 2NH3 = 1 × 102 [N2] [H2]3 - 1 × 10-3 [NH3]2 and at some instant if = 10-5 M2t...The rate equation for the reaction 2A + B → C is found to be: rate k[A][B]. The correct statement in relation to t...What is ᐃH for the reaction A + B → C, where the mechanism involves several kinetic steps....