Chemical Kinetics and Nuclear ChemistryHard
Question
Number of natural life times (Tav) required for a first-order reaction to achieve 99.9% level of completion is :
Options
A.2.3
B.6.9
C.9.2
D.0.105
Solution
We know
k =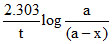
99.9% completion
a = 100
a × x = (100 - 99.90) = .10
Then t =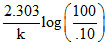
t = 2.303 × 3 ×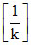
t = 6.9 × tav
k =
99.9% completion
a = 100
a × x = (100 - 99.90) = .10
Then t =
t = 2.303 × 3 ×
t = 6.9 × tav
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
A radioactive element has a half life of one day. After three days the amount of the element left will be :...Bombardment of aluminium by a-particle leads to its artificial disintegration in two ways, (i) and (ii) as shown. Produc...For the complex Ag+ + 2NH3 ⇋ [Ag(NH3)2+] = 2 × 107 L2 mol-2 s-1 [Ag+] [NH3]2 - 1 × 10-2 s-1 [Ag(NH3)2+]H...Table I (reactions) and table II (methods) have been matchedCorrect matching is :...In a bimolecular reaction, the steric factor P was experimentally determined to be 4.5. The correct option(s) among the ...