Chemical Kinetics and Nuclear ChemistryHard
Question
Which of the following statement are true regarding the log K vs.1/T plot shown in the given diagram ?
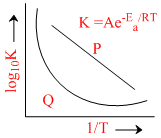

Options
A.Plot P shows that the energy of activation is independent of temperature.
B.Plot Q describes the behaviour of temperature dependence of energy of activation
C.Arrhenius behaviour is described by P
D.The slope of curve P gives the value - 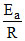
Solution
Use K = Ae- Ea/RT
and lnk = lnA =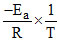
2.303 log K = 2.303 log A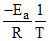
log K = log A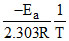
and lnk = lnA =
2.303 log K = 2.303 log A
log K = log A
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Penetrating power of α-particle is :...The increasing order of basicity of the following compounds is : (A)Â Â Â Â Â (B)Â Â (C)Â Â Â Â Â Â (D)Â...Mechanism of a hypothetical reaction X2 + Y2 → 2XY is given below : (i) X2 → X + X(fast) (ii) X + Y2 ⇌...For the complex Ag+ + 2NH3 ⇋ [Ag(NH3)2+] = 2 × 107 L2 mol-2 s-1 [Ag+] [NH3]2 - 1 × 10-2 s-1 [Ag(NH3)2+]H...Half life period of 10 gm radioactive element is 20 days. Then half life period of 100 gm of this element is :...