Chemical Kinetics and Nuclear ChemistryHard
Question
Rate constant k varies with temperature by equation , log k(min-1) = 5 - 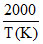 . We can conclude:
. We can conclude:
Options
A.pre-exponential factor A is 5
B.Ea is 2000 kcal
C.pre-exponential factor A is 105
D.Ea is 9.212 kcal
Solution
(C) Given, log k (min-1) = 5 - 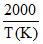
Compare this with
log K = log A -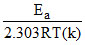
we find A = 1 × 105
(D)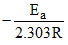 = - 2000
= - 2000
Ea = 9.212 k cal.
Compare this with
log K = log A -
we find A = 1 × 105
(D)
Ea = 9.212 k cal.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Select correct statement (s) :...The correct order of N-compounds in its decreasing order of oxidation states is...The rate law for a reaction between the substances A and B is given by rate = K[A]n [B]m. On doubling the concentration ...Which of the following is incorrect about freundlich adsorption isotherm ?...Consider the chemical reaction,N2(g) + 2H2(g) → 2NH3(g) The rate of this reaction can be expressed in terms of tim...