Chemical Kinetics and Nuclear ChemistryHard
Question
In the following reaction, how is the rate of appearance of the underlined product related to rate of disappearance of the underlined reactant :
BrO3- (aq) + 5Br- (aq) + 6H+ (aq) 3Br2 (l) + 3H2O (l)
BrO3- (aq) + 5Br- (aq) + 6H+ (aq) 3Br2 (l) + 3H2O (l)
Options
A.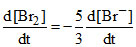
B.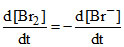
C.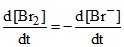
D.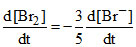
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
A radioactive element gets spilled over the floor of a room. Its half-life period is 30 days. If the initial activity is...The addition of a catalyst during a chemical reaction alters which of the following quantities?...The decomposition of N2O5 according to the equation 2N2O5(g) → 4NO2(g) + O2(g) is a first order reaction. After 30 minut...The rate of chemical reaction is directly proportional to :...For a first order reaction A(g) → 2B(g) + C(g) at constant volume and 300 K, the total pressure at the beginning (...