JEE Advanced | 2018Chemical Kinetics and Nuclear ChemistryHard
Question
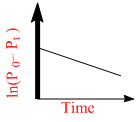
For a first order reaction A(g) 2B(g) + C(g) at constant volume and 300 K, the total pressure at the beginning (t = 0) and at time t are P0 and Pt, respectively. Initially, only A is present with concentration [A]0, and t1/3 is the time required for the partial pressure of A to reach 1/3rd of its initial value. The correct option(s) is (are) :-
(Assume that all these gases behave as ideal gases)
Options
A.
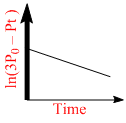
B.
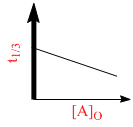
C.
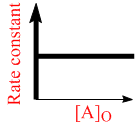
D.
Solution
A2B + C
t = 0 P0 – –
t = t P0–P 2P P
P0 + 2P = Pt
Rate constant does not depends on concentration
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The compound A decomposed (A → product) by first order kinetics. If the concentration of A dropped from 0.1 M to 0...In a consecutive reaction system $A\overset{\quad E_{1}\quad}{\rightarrow}B\overset{\quad E_{2}\quad}{\rightarrow}C$ whe...For the reaction : H2 = Cl2 2HClthe order of reaction is...For the chemical reaction A + 2B → C, rate of the reaction is increased by 2.82 times when the concentration of A ...For the second-order reaction 2A → B, time taken for the [A] to fall to one-fourth value is how many times the time it t...