Chemical Kinetics and Nuclear ChemistryHard
Question
If 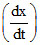 = k [H+]n and rate becomes 100 times when pH changes from 2 to 1. Hence, order is :
= k [H+]n and rate becomes 100 times when pH changes from 2 to 1. Hence, order is :
Options
A.1
B.2
C.3
D.0
Solution
Given, pH change from 2 to 1
Then H+ concentration change from 10-2 M to 10-1 M
Then
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The rate of diffusion of a gas having molecular weight just double of N2 gas is 56 mL/sec. The rate of diffusion of N2 g...The reaction of NO2 (g) and O3 (g) is first-order in NO2 (g) and O3 (g) 2NO2 (g) + O3 (g) → N2O5 (g) + O2 (g)The r...The compound used in enrichment of uranium for nuclear power plant is :...The colligative property is not represented by :...The half life period of a first order chemical reaction is 6.93 minutes. The time required for the completion of 99% of ...