Chemical Kinetics and Nuclear ChemistryHard
Question
The reaction of NO2 (g) and O3 (g) is first-order in NO2 (g) and O3 (g)
2NO2 (g) + O3 (g) → N2O5 (g) + O2 (g)
The reaction can take place by mechanism :
I : NO2 + O3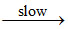 NO3 + O2
NO3 + O2
NO3 + NO2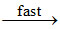 N2O5
N2O5
II : O3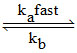 O2 + [O]
O2 + [O]
NO2 + O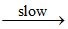 NO3
NO3
NO3 + NO2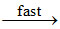 N2O5
N2O5
Select correct mechanism.
2NO2 (g) + O3 (g) → N2O5 (g) + O2 (g)
The reaction can take place by mechanism :
I : NO2 + O3
NO3 + NO2
II : O3
NO2 + O
NO3 + NO2
Select correct mechanism.
Options
A.I only
B.II only
C.both I and II
D.None of I and II
Solution
For Rxn rate determining step is slowest step
Then in 1st Rxn
Rate = k [NO2] [O3] .....(i)
But 2nd Rxn
O3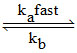 O2 + [O] ....(a)
O2 + [O] ....(a)
NO2 + O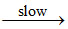 NO3 ....(b)
NO3 ....(b)
NO3 + NO2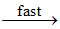 N2O5 ....(c)
N2O5 ....(c)
Then for Rxn (a)
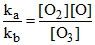 = keq ....(d)
= keq ....(d)
by Rxn (b)
Rate = k [NO2] [O] ....(ii)
put value of [O] from (d) to (ii)
Rate = k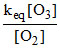 × [NO2]
× [NO2]
Rate =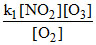
Then in 1st Rxn
Rate = k [NO2] [O3] .....(i)
But 2nd Rxn
O3
NO2 + O
NO3 + NO2
Then for Rxn (a)
by Rxn (b)
Rate = k [NO2] [O] ....(ii)
put value of [O] from (d) to (ii)
Rate = k
Rate =
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Aqueous ammonia can be used to separate which of the following pair of ions ?...For the reaction : H2 = Cl2 2HClthe order of reaction is...At 325°C, 1,3-butadiene dimerizes according to the equation: 2C4H6(g) → C8H12(g). It is found that the partial pressure ...The bromination of acetone that occurs in acid solution is represented by CH3COCH3 (aq) + Br2 (aq) → CH3COCH2 Br (...Isomers of hexane, based on their branching, can be divided into three distinct classes as shown in the figure. The corr...