Chemical Kinetics and Nuclear ChemistryHard
Question
The rate constant of a reaction is given by k = 2.1 × 1010 exp(-2700/RT). It means that
Options
A. log k versus 1/T will be a straight line with slope = 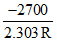
B.log k versus 1/T will be a straight line with intercept on log k axis = 2.1 × 1010.
C.The number of effective collisions are 2.1 × 1010cm-3s-1.
D.Half life of the reaction increases with increase of temperature.
Solution
For Arrhenius equation k = Ae-Ea/RT
= 2.1 × 1010 × e-2700/RT
or
log k = log A -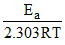 = log 2.1 × 1010 -
= log 2.1 × 1010 - 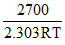
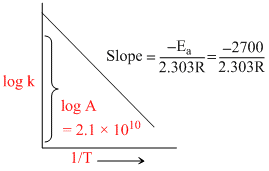
Plot of log k vs 1/T
= 2.1 × 1010 × e-2700/RT
or
log k = log A -
Plot of log k vs 1/T

Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The rate law for the dimerisation of NO2 is = k [NO2]2which of the following changes will change the value of the specif...The rate of reaction A + B → Products is given by the equation r = k[A][B]. If B is taken in large excess, the ord...For the non-equilibrium process, A + B → products, the rate is first-order w.r.t. A and second-order w.r.t. B. If ...α - Particles can be detected using...If no catalyst (H+) is present in acid hydrolysis of ester (in above question) then rate constant K is :...