Chemical Kinetics and Nuclear ChemistryHard
Question
Consider following reaction,
Zn + 2H+ → Zn2+ + H2
Half-life period is independent of concentration of Zn at constant pH. At constant Zn concentration, half-life is 10 minutes at pH = 2 and half-life is 100 minutes at pH = 3. Hence, rate law is :
Zn + 2H+ → Zn2+ + H2
Half-life period is independent of concentration of Zn at constant pH. At constant Zn concentration, half-life is 10 minutes at pH = 2 and half-life is 100 minutes at pH = 3. Hence, rate law is :
Options
A. k [Zn][H+]
B.k [Zn][H+]2
C.k [Zn]0 [H+]
D.k [Zn]0 [H+]2
Solution
Half life period independent on conc.hence order is unity wrt Zn
Let order with respect to H+ ions is ′a′
Then the rate law will be Rate = k [Zn][H+]a
At pH = 2, [H+] = 10-2 So the rate law becomes Rate = (k[10-2]a)[Zn] = k1 [Zn]
so the half life will be T1/2 =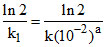 = 10 min
= 10 min
At pH = 3, [H+] = 10-3 So the rate law becomes Rate = (k[10-3]a )[Zn] = k2 [Zn]
so the half life will be T1/2 =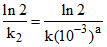 = 100 min
= 100 min
so we have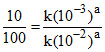
which will give 10-1 = 10-a
Hence a = 1
Let order with respect to H+ ions is ′a′
Then the rate law will be Rate = k [Zn][H+]a
At pH = 2, [H+] = 10-2 So the rate law becomes Rate = (k[10-2]a)[Zn] = k1 [Zn]
so the half life will be T1/2 =
At pH = 3, [H+] = 10-3 So the rate law becomes Rate = (k[10-3]a )[Zn] = k2 [Zn]
so the half life will be T1/2 =
so we have
which will give 10-1 = 10-a
Hence a = 1
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Arrange following compounds in decreasing order of reactivity for hydrolysis reaction :-(I) C6H5COCl (II) NO2 COCl(III) ...Select the incorrect statement from the following....For a process A+ B Products, the rate is first order with respect to A and First order with respect to B. When 1.0 mol e...Which of the following is correct about first-order reaction?...A zero-order reaction is one...