Chemical Kinetics and Nuclear ChemistryHard
Question
Acid catalysed hydrolysis of ester is first-order reaction and rate constant is given by
k =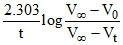
where V0, Vt and V∞ are the volume of standard NaOH required to neutralise acid present at a given time ; if ester is 50% hydrolysed then :
k =
where V0, Vt and V∞ are the volume of standard NaOH required to neutralise acid present at a given time ; if ester is 50% hydrolysed then :
Options
A.V∞ = vt
B.V = (Vt - V0)
C.V∞ = 2Vt - V0
D.V∞ = 2Vt + V0
Solution
If easter 50% hydrolysed then
x =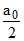
(a0 - x) =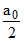
We can write
a0 ∞ (V∞ - V0)
(a0 - x) ∞ (V∞ - Vt)
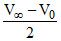 = (V∞ - Vt)
= (V∞ - Vt)
V∞ - V0 = 2V∞ - 2Vt
Vt =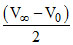
or V∞ = 2 Vt - V0
x =
(a0 - x) =
We can write
a0 ∞ (V∞ - V0)
(a0 - x) ∞ (V∞ - Vt)
V∞ - V0 = 2V∞ - 2Vt
Vt =
or V∞ = 2 Vt - V0
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Graph between log k and 1/T (k is rate constant in s-1 and T is the temperature in K) is a straight line. As shown in fi...6C12 and 1T3 are formed in nature due to the nuclear reaction of neutron with...Decay constant of a radioactive substance is 69.3sec-1, find t1/16 of the same substance....In a zero-order reaction for every 10o rise of temperature, the rate is doubled. If the temperature is increased from 10...Sulphide ores of metals are usually concentrated by froth floatation process. Which one of the following sulphide ores i...