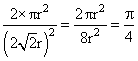Solid StateHard
Question
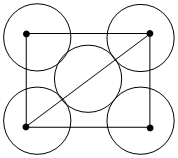
The packing efficiency of the two-dimensional square unit cell shown below is

Options
A.39.27%
B.68.02%
C.74.05%
D.78.54%
More Solid State Questions
The ionic radii of Rb+ and I− are 1.46 and 2.16 Å, respectively. The most probable type of structure exhibited by it is...NaCl is doped with 2 × 10–3 mole% SrCl2, the concentration of cation vacancies is (NA = 6 × 1023)...In which of the following crystals, alternate tetrahedral voids are occupied?...Which of the following shaded plane in fcc lattice contains arrangement of atoms as shown by circles :...In a face centred cubic lattice, atom A occupies the corner positions and atom B occupies the face centre positions. If ...