Solid StateHard
Question
In a face centred cubic lattice, atom A occupies the corner positions and atom B occupies the face centre positions. If one atom of B is missing from one of the face centred points, the formula of the compound is
Options
A.AB2
B.A2B3
C.A2B5
D.A2B
Solution
Effective no.of A atoms = 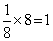
Effective no.of B atoms =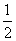 × 5 (One is missing) =
× 5 (One is missing) = 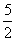
Therefore formula is A1B5/2 = A2B5
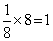
Effective no.of B atoms =
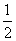 × 5 (One is missing) =
× 5 (One is missing) = 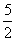
Therefore formula is A1B5/2 = A2B5
Create a free account to view solution
View Solution FreeMore Solid State Questions
In the zinc blend structure (ZnS), S2− adopts CCP arrangement and Zn2+ occupies...A binary solid (A+ B−) has a rock salt structure. If the edge length is 400 pm and the radius of cation is 75 pm, then t...Which of the following are the correct axial distance and axial angles for rhombohedral system?...Which of the following is/are correct statement(s) about Fe3O4 crystal?...The total number of 2nd nearest Cl- ions to the Na+ ion in NaCl unit cell are -...