ElectrochemistryHard
Question
In which of the following cell (s) : Ecell = Eocell ?
Options
A.Cu(s)|Cu2+(0.0 1M)||Ag+ (0.1M)|Ag(s)
B.Pt(H2)|pH = 1|Zn2+ (0.01M)|Zn(s)|
C.Pt(H2)|pH = 1|Zn2+ (1M)|Zn(s)|
D.Pt(H2)|H+ = 0.1M||Zn2+ (0.01M)|Zn(s)
Solution
(A) ECell = EoCell - 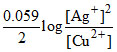
(B) ECell = EoCell -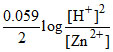
(D) ECell = EoCell -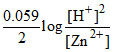
(B) ECell = EoCell -
(D) ECell = EoCell -
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Zn Amalgam is prepared by electrolysis of aqueous ZnCl2 using Hg cathode (9gm). How much current is to be passed through...An acidic solution of copper (II) sulphate containing some contaminations of zinc and iron (II) ions was electrolysed ti...Select the correct statement.(i) Delocalisation of σ-electron is hyperconjugation.(ii) Delocalisation of π-el...The standard reduction potentials at 298 K for the following half reactions are given against each : Zn2+(aq) + 2e- V...How much the potential of a hydrogen electrode will change when its solution initially at pH = 0 is neutralised to pH = ...