ElectrochemistryHard
Question
How much the potential of a hydrogen electrode will change when its solution initially at pH = 0 is neutralised to pH = 7 ?
Options
A.Increase by 0.059 V
B.Decrease by 0.059 V
C.Increase by 0.41 V
D.Decrease by 0.41 V
Solution
H+ + e- → 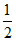 H2. E = 0 -
H2. E = 0 - 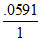 log10
log10 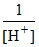 = + 0.591 log10[H+].
= + 0.591 log10[H+].
E1 = 0 {pH = 0}.
E2 = + 0.0591log10[10-7] = - .0591 × 7 {at pH = 7} = - 0.41 V.
E1 = 0 {pH = 0}.
E2 = + 0.0591log10[10-7] = - .0591 × 7 {at pH = 7} = - 0.41 V.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Consider the following statements:(I) is more stable than (II) is more stable than (III) is more stable than (IV) is mor...Any redox reaction would occur spontaneously, if :...Pt | (H2) | pH = 1 || pH = 2 | (H2)Pt 1 atm 1 atmThe cell reaction for the given cell is :...Calculate the ionic product of water at 25°C from the following data:Conductivity of water = 5.5 × 10−6 mho m−1$\lambda_...In salt bridge, normally KCl is used because...