ElectrochemistryHard
Question
M(s) → Mn+ (aq) + ne-
Choose the correct statement(s).
Choose the correct statement(s).
Options
A.EM | Mn+ decreases with increase in [Mn+]
B.E Mn+ | M increases on increasing temperature.
C.E Mn+ | M increases on increasing [Mn+]
D.EM | Mn+ increases on increasing temperature.
Solution
EM | M+n = EoM/M+n - 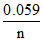 log Mn+
log Mn+
[Mn+] ↑, EM/Mn+ ↑, EMn+/M ↑.
[Mn+] ↑, EM/Mn+ ↑, EMn+/M ↑.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Given standard electrode potentials : Fe3+ + 3e- → Fe ; Eo = - 0.036 voltFe2+ + 2e- → Fe; Eo = - 0.440 volt ...At 298K the standard free energy of formation of H2O(l) is -237.20 kJ/mole while ᐃGo of its ionisation into H+ ion...The standard reduction potential of oxygen in acidic solution is +1.23 V. What is the standard reduction potential of ox...The correct relationship between Gibb′s energy change in a reaction and the corresponding equilibrium constant KC ...Equal volumes of 0.015 M – CH3COOH and 0.015 M – NaOH solutions are mixed together. What would be the molar conductivity...