ElectrochemistryHard
Question
A current of 2 A was passed for 1 h through a solution of CuSO4 0.237g of Cu2+ ions were discharged at cathode. The current efficiency is
Options
A.42.2%
B.26.1%
C.10%
D.40.01%
Solution
W = 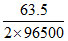 × 2 × 60 × 60 = 2.37 gm
× 2 × 60 × 60 = 2.37 gm
% of efficiency =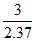 × 100.
× 100.
% of efficiency =
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
For the reduction of NO3- ion in an aqueous solution, Eo is +0.96 V. Values of Eo for some metal ions are given below V2...During electrolysis of water the volume of O2 Liberated is 2.24 dm3. The volume of hydrogen liberated, under same condit...In a conductivity cell, the two platinum electrodes, each of area 10 cm2 are fixed 1.5 cm apart. The cell contained 0.05...At what pH does the potential (EMF) for the disproportionation of chlorine change from a negative value to a positive va...These are three canonical structures of naphthalene. Examine them and find correct statement among the following :...