ElectrochemistryHard
Question
Which of following cell can produce more electrical work.
Options
A.Pt, H2 |0.1M NH4Cl || 0.1 M CH3COOH | H2, Pt
B.Pt, H2 | 0.1 M HCl || 0.1 M NaOH | H2, Pt
C.Pt, H2 | 0.1 M HCl || 0.1 M CH3COOK | H2, Pt
D.Pt, H2 | 0.1 M CH3COOK || 0.1 M HCl | H2, Pt
Solution
Ecell = - 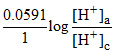
For Ecell to be highest [H+]a should be lower and [H+]c should be higher and that why anode compartment should be more basic and cathodic compartment should be acidic.
For Ecell to be highest [H+]a should be lower and [H+]c should be higher and that why anode compartment should be more basic and cathodic compartment should be acidic.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A 0.5 M NaOH solution offers a resistance of 31.6 ohm in a conducivity cell at roomtemperature. What shall be the appori...Which of the following molecule contains contains one lone pair of electrons on the central atom?...During electrolysis of water the volume of O2 Liberated is 2.24 dm3. The volume of hydrogen liberated, under same condit...Which cannot displace hydrogen from its compound ?...When the sample of copper with zinc impurity is to be purified by electolysis, the appropriate electrode are...