ElectrochemistryHard
Question
At equimolar concentrations of Fe2+ and Fe3+, what must [Ag+] be so that the voltage of the galvanic cell made from the (Ag+ | Ag) and (Fe3+ | Fe2+) electrodes equals zero ?
Fe2+ + Ag+ ⇋ Fe3+ + Ag
EoAg+||Ag = 0.7991 ; EoFe2+|Fe2+ = 0.771
Fe2+ + Ag+ ⇋ Fe3+ + Ag
EoAg+||Ag = 0.7991 ; EoFe2+|Fe2+ = 0.771
Options
A.0.34
B.0.44
C.0.47
D.0.61
Solution
0 = (-0.771 + 0.7991) - 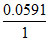 log
log 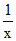 ⇒ 0 = 0.0281 + 0.0591 log X
⇒ 0 = 0.0281 + 0.0591 log X
log X = -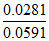 ⇒ X = 0.335 M
⇒ X = 0.335 M
log X = -
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
When 12,000 coulombs of electricity is passed through the electrolyte, 3.0 g of a metal of atomic mass 96.5 g/mol is dep...Consider the following reduction processes :$${{Al}^{3 +} + 3e^{-} \rightarrow Al(s),E^{\circ} = - 1.66\text{ }V }{{Fe}^...The conductivity of saturated solution of Ba3(PO4)2 is 1.2 × 10−5 Ω−1 cm−1. The limiting equivalent conductivities of Ba...ᐃG is the available energy (energy produced) during the electrochemical reaction in galvanic cell which can be con...When silver chloride is dissolved in a large excess of ammonia, practically all silver ion can be assumed to exist in fo...