ElectrochemistryHard
Question
The first ionisation potential of Na is 5.1 eV. The value of electron gain enthalpy of Na+ will be :-
Options
A.- 2.55 eV
B.- 5.1 eV
C.- 10.2 eV
D.+ 2.55 eV
Solution
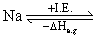 Na+ + e-
Na+ + e- |I.E.| = |ᐃHe.g.| Hence
ᐃHe.g. of Na+ = - 5.1 eV
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
If φ denotes reduction potential, then which is true ?...A current of 9.65 ampere is passed through the aqueous solution NaCl using suitable electrodes for 1000 s. The amount of...The reaction Zn(s) + 2AgCl(g) → ZnCl2(aq) + 2Ag(s) occurs in the cell Zn |ZnCl2 (1M solution), AgCl(s) | Ag. The n...The standard emf of the cell, Cd(s) | CdCl2(aq) (0.1 M) || AgCl(s) | Ag(s) in which the cell reaction is,Cd(s) + 2AgCl(s...The emf of Daniel cell at 298 K is E1Zn|ZnSO4(0.01M)||CuSO4(1.0M)|Cu When the concentration of ZnSo4 is 1.0 M and that o...