ElectrochemistryHard
Question
A cell Cu | Cu++ || Ag+ | Ag initially contains 2M Ag+ and 2M Cu++ ions in 1L electrolyte. The change in cell potential after the passage of 10 amp current for 4825 sec is :
Options
A.- 0.00738 V
B.- 1.00738 V
C.- 1.00738 V - 0.0038 V
D.none
Solution
Q = 10 × 4825 = 48250 C
no. of faraday =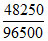 = 0.5
= 0.5
Ag + Cu++ → Ag+ +
Cu++ → Ag+ +  Cu
Cu
2.00 2.00
2-0.25 2 + 0.50
Ecell = EoCell -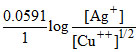
E1 = EoCell -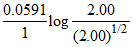
E2 = EoCell -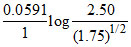
ᐃE = E2 - E1 =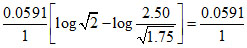 [log 1.41 - log 1.88]
[log 1.41 - log 1.88]
=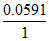 [0.1492 - 0.2742] = -
[0.1492 - 0.2742] = - 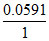 × 0.125 = - 0.00738 V.
× 0.125 = - 0.00738 V.
no. of faraday =
Ag +
2.00 2.00
2-0.25 2 + 0.50
Ecell = EoCell -
E1 = EoCell -
E2 = EoCell -
ᐃE = E2 - E1 =
=
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The value of equilibrium constant for a feasible cell reaction must be...Which statement is correct....Given the data at 25oC, Ag + I- → Agl + e- ; Eo = 0.152 V Ag → Ag+ + e- : Eo = - 0.800 V What is the value o...From an electrolyte, one mole of electron will deposit at cathode...MnO4- + 8H+ + 5e- → Mn2+ + 4H2O, If H+ concentration is decreased from 1 M to 10-4 M at 25oC, where as concentrati...