ElectrochemistryHard
Question
MnO4- + 8H+ + 5e- → Mn2+ + 4H2O,
If H+ concentration is decreased from 1 M to 10-4 M at 25oC, where as concentration of Mn2+ and MnO4- remain 1 M.
If H+ concentration is decreased from 1 M to 10-4 M at 25oC, where as concentration of Mn2+ and MnO4- remain 1 M.
Options
A.the potential decreases by 0.38 V with decrease in oxidising power
B.the potential increases by 0.38 V with increase in oxidising power
C.the potential decreases by 0.25 V with decrease in oxidising power
D.the potential decreases by 0.38 V without affecting oxidising power
Solution
MnO4- + 8H+ + 5e- → Mn2+ + 4H2O
E1 = Eo -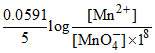
E2 = Eo -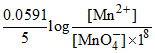 = -
= - 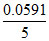 × 32 = - 0.37824
× 32 = - 0.37824
E1 - E2 = 0.38 Volt.
E1 = Eo -
E2 = Eo -
E1 - E2 = 0.38 Volt.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The conductivity of saturated solution of Ba3(PO4)2 is 1.2 × 10−5 Ω−1 cm−1. The limiting equivalent conductivities of Ba...The limiting molar conductivities ∧o for NaCl, KBr and KCl are 126, 152 and 150 S cm2 mol-1 respectively. The W...During the electrolysis of 0.1 M CuSO4 solution using copper electrodes, a depletion of [Cu++] occurs near the cathode w...A metal having negative reduction potential, when dipped in the solution of its own ions, has a tendency to...In hydrogen - oxygen fuel cell, combustion of hydrogen occurs to...