ElectrochemistryHard
Question
Which one of the following will increase the voltage of the cell ? (T = 298 K)
Sn + 2Ag+ → Sn2+ + 2Ag
Sn + 2Ag+ → Sn2+ + 2Ag
Options
A.increase in the size of silver rod
B.increase in the concentration of Sn+2 ions
C.increase in the concentration of Ag+ ions
D.none of the above
Solution
Ecell = Eocell - 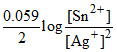 Ag+ increase, Ecell increase.
Ag+ increase, Ecell increase.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
To perform an analysis of a mixture of metal ions by electro-deposition, the second metal to be deposited must not being...The two aqueous solutions; A (AgNO3) and B(LiCl) were electrolysed using Pt. electrodes. The pH of the resulting solutio...Choose the correct statement(s)....The function(s) of salt bridge in a cell is/ are...Given below are the half-cell reactions Mn2+ + 2e- → Mn ; Eo = - 1.18 V (Mn3+ + e- → Mn2+) ; Eo = + 1.51 V T...