ElectrochemistryHard
Question
A current of 0.1A was passed for 2hr through a solution of cuprocyanide and 0.3745 g of copper was deposited on the cathode. Calculate the current efficiency for the copper deposition. (Cu - 63.5)
Options
A.79%
B.39.5 %
C.63.25%
D.63.5%
Solution
m (theoretical) = 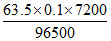 = 0.4738 g
= 0.4738 g
∴ % efficiency =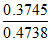 × 100 = 79 %
× 100 = 79 %
∴ % efficiency =
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
At 25°C, the solubility product of Pb(OH)2 (s) is [Given: $E_{Pb^{2 +}|Pb}^{o} = - 0.13\text{ V;}E_{OH^{-}|Pb(OH)_{2}|Pb...Assertion :- BeSO4 is more soluble in water than BaSO4.Reason :- In BeSO4 H.E. exceeds its L.E....Which of the following has longest C - O bond :...Which of the following steyements is true for the electrochemical Daniel cell?...When an electric current is passed through an aqueous solution of the following, the concentrations of cation as well as...