ElectrochemistryHard
Question
The oxidation number of sulphur in H2S2O7 is :
Options
A.+ 2
B.+ 6
C.+ 4
D.+ 8
Solution
Oxidation number of sulphur in H2S2O7 can be calculated as follows: 2 + 2x - 14 = 0
2x = 14 - 2 = 12
x =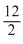 = 6
= 6
2x = 14 - 2 = 12
x =
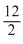 = 6
= 6Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
At what pH does the potential (EMF) for the disproportionation of chlorine change from a negative value to a positive va...Which of the following curve represents the variation of λM with √C for AgNO3 ?...Assuming that hydrogen behaves as an ideal gas, what is the EMF of the cell at 25°C if P1 = 600 mm and P2 = 420 mm: Pt |...Any redox reaction would occur spontaneously, if :...A gas ‘X’ at 1 atm is bubbled through a solution containing a mixture of 1 M – Y− and 1 M – Z− at 25°C. If the reduction...