ElectrochemistryHard
Question
Zn | Zn2+ (C1)|| Zn2+ (C2)|Zn. for this cell ᐃG is negative if -
Options
A.C1 = C2
B.C1 > C2
C.C2 > C1
D.None
Solution
Zn → Zn2+(C1) + 2e-
Zn2+(C2) + 2e- → Zn
Zn2+(C2) ⇋ Zn2+(C1)
E = 0 -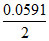 log
log 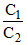
E → +ve When C1 < C2
Zn2+(C2) + 2e- → Zn
Zn2+(C2) ⇋ Zn2+(C1)
E = 0 -
E → +ve When C1 < C2
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Oxidation number of Os in OsO4:...The specific conductance of a N/10 KCl at 25oC is 0.0112 ohm-1 cm-1. The resistance of cell containing solution at the s...In which of the following molecules π - electron density in ring is minimum :...Which is/are correct among the following ?Given, the half cell emf′s EoCu+2|Cu = 0.337, EoCu+1|Cu = 0.521...A current of 2 A was passed for 1 h through a solution of CuSO4 0.237g of Cu2+ ions were discharged at cathode. The curr...