ElectrochemistryHard
Question
Eo for F2 + 2e → 2F- is 2.8 V, Eo for 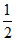 F2 + e → F- is
F2 + e → F- is
Options
A.2.8 V
B.1.4 V
C.- 2.8 V
D.- 1.4 V
Solution
Eo is intensive property and it do not depend on mass of F2 taking part.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Although aluminium is above hydrogen in the electrochemical series, it is stable in air and water. Why?...Which of the following molecule contains contains one lone pair of electrons on the central atom?...Anthracene, C14H10, can be oxidized to anthraquinone, C14H8O2. What weight of anthraquinone can be produced by the passa...The chemical reaction,2AgCl(s) + H2(g) → 2HCl(aq) = 2Ag(s)Taking place in a galvanic cell is represented by the no...When silver chloride is dissolved in a large excess of ammonia, practically all silver ion can be assumed to exist in fo...