SolutionHard
Question
For an ideal binary solution with PA0 / PB0 which relation between XA (mole fraction of A in liquid phase) and YA (mole fraction of A in vapour phase) is correct , XB and YB are mole fraction of B in liquid and vapour phase respectively : (Given : PA0 > PB0)
Options
A.XA = YA
B.XA > YA
C.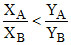
D.XA, YA, XB and YB cannot be correlated
Solution
Mole fraction of more volatile substance is greater in vapour phase.
Create a free account to view solution
View Solution FreeMore Solution Questions
Which of the following forms ideal solution ?...Van′t Hoff factors of aqueous solutions of X, Y, Z are 1.8, 0.8 and 2.5. Hence, their (assume equal concentrations...At 323 K, the vapour pressure in millimeters of mercury of a methanol-ethanol solution is represented by the equation p ...The vapour pressure of a dilute solution of a non-volatile solute is not influenced by the...A complex of iron and cyanide ions is 100% ionised at 1m (molal). If its elevation in b.p. is 2.08. Then the complex is ...