SolutionHard
Question
Water and chlorobenzene are immiscible liquids. Their mixture boils at 90oC under a reduced pressure of 7.82 × 104 Pa. The vapour pressure of pure water at 90oC is 7.03 × 104 Pa. On weight percent basis, chlorobenzene in the distillate is equal to (mol. wt. of chlorobenzene is 112.5 g mol-1)
Options
A.50
B.60
C.70
D.80
Solution
PV = nRT
Pmix = 7.82 × 104 Pa
P2 + P1 = 7.82 × 104 Pa
P1 = 7.03 × 104 Pa
∴ P2 = 0.79 × 104 Pa
P ∞ n
∴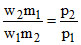
w2 = w1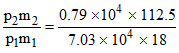 = 0.70
= 0.70
Pmix = 7.82 × 104 Pa
P2 + P1 = 7.82 × 104 Pa
P1 = 7.03 × 104 Pa
∴ P2 = 0.79 × 104 Pa
P ∞ n
∴
w2 = w1
Create a free account to view solution
View Solution FreeMore Solution Questions
When 20 g of naphthoic acid (C11H8O2) is dissolved in 50 g of benzene (Kf = 1.72 K kg mol-1), a freezing point depressio...Which represents correct difference ?I. II. III....At 80oC, the vapour pressure of pure liquid ′A′ is 520 mm Hg and that of pure liquid ′B′ is 1000...Consider the following four liquids :1. Water : highly polar ; H-bonding2. Hexanol : slightly polar ; some H-bonding3. C...Which characterises the weak intermolecular forces of attraction in a liquid ?...