SolutionHard
Question
What is the normal freezing point of the solution represented by the phase diagram ?
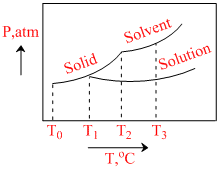

Options
A.T1
B.T2
C.T3
D.T0
Solution
From given graph, we can say T1 is that temp at which solid state and liquid (solution) are in equilibrium.
Create a free account to view solution
View Solution FreeMore Solution Questions
Barium ions, CN- and Co2+ form an ionic complex. If that is supposed to be 75% ionised in water with vant Hoff factor (i...One mole of a solute A is dissolved in a given volume of a solvent. The association of the solute take place as follows ...For 1 molal aqueous solution of the following compounds, which one will show the highest freezing point ?...A binary liquid solution is prepared by mixing n-heptane and ethanol. Which one of the following statements is correct r...At higher altitudes, water boils at temperature < 100oC because...