SolutionHard
Question
Barium ions, CN- and Co2+ form an ionic complex. If that is supposed to be 75% ionised in water with vant Hoff factor (i) equal to four, then the coordination numbers of Co2+ in the complex can be:
Options
A.6
B.4
C.6 & 4 both possible
D.5
Solution
α = 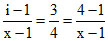 ∴ x = 5 ∴ Complex is Ba3[Co(CN)5]2
∴ x = 5 ∴ Complex is Ba3[Co(CN)5]2
Create a free account to view solution
View Solution FreeMore Solution Questions
Fog is a colloidal solution of:...pressure cooker reduces cooking time for food because...Kf for water is 1.86 K Kgmol-1. If your automobile radiator holds 1 kg water, how many grams of ethylene glycol (C2H6O2)...Which of the following is less than zero for ideal solutions ?...For an ideal binary solution with PA0 / PB0 which relation between XA (mole fraction of A in liquid phase) and YA (mole ...