SolutionHard
Question
Solubility curves of four ionic salts X, Y, Z, W are given below :
In which case the value of ᐃHsol. < 0
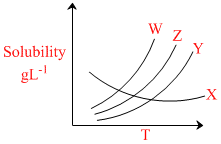
In which case the value of ᐃHsol. < 0

Options
A.X
B.Y
C.Z
D.W
Solution
ᐃHsolution < 0 ; Exothermic
Temperature ↑; Solubility ↓
for X, Temperature ↑; Solubility ↓
Temperature ↑; Solubility ↓
for X, Temperature ↑; Solubility ↓
Create a free account to view solution
View Solution FreeMore Solution Questions
A certain compound (X) is used in laboratory for analysis, its aqueous solution gives brown appearanceon addition copper...What is the normal boiling point of the solution represented by the phase diagram ?...What will be the molecular weight of NaCl determined experimentally from elevation in the boiling point or depression in...Which one of the following statements is correct when SO2 is passed through acidified K2Cr2O7 solution?...Osmotic pressure of blood is 7.40 atm at 27oC. Number of mol of glucose to be used per L for an intravenous injection th...