SolutionHard
Question
Osmotic pressure of blood is 7.40 atm at 27oC. Number of mol of glucose to be used per L for an intravenous injection that is to have the same osmotic pressure as blood is :
Options
A.0.3
B.0.2
C.0.1
D.0.4
Solution
π = CRT 7.40 = n × 0.0821 × 300
π =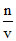 RT n =
RT n = 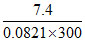 = 0.3.
= 0.3.
π =
Create a free account to view solution
View Solution FreeMore Solution Questions
Which has the equal boiling point ?...Which is/are correct statement(s) ?...What is pH of the solution when 100 ml of 0.1M HCl is mixed with 100 ml of 0.1M CH3COOH....Maximum osmotic pressure will be shown by which of the following solution (all 90% ionized)?a) 0.1 M-AlCl3 (b) 0.1 M-BaC...In mixture A and B component show - ve deviation as...