SolutionHard
Question
All of the water in a 0.20 M solution of NaCl was evaporated and a 0.150 mol of NaCl was obtained. What was the original volume of the sample ?
Options
A.30 mL
B.333 mL
C.750 mL
D.1000 mL
Solution
Let Vml be the original volume of the smaple,
∴ 0.20 =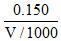 V = 750 ml.
V = 750 ml.
∴ 0.20 =
Create a free account to view solution
View Solution FreeMore Solution Questions
The vapour pressure of pure benzene at 88°C is 960 mm and that of toluene at the same temperature is 380 mm of benzene. ...Temperature does not affect :...Which of the following aqueous solution will have the highest boiling point?...A 5.25 % solution of a substance is isotonic with a 1.5% solution of urea (molar mass = 60 g mol-1) in the same solvent....Consider following solutions :I : 1 M aqueous glucose solution II : 1M aqueous sodium chloride solution III : 1M aqueous...