Atomic StructureHard
Question
Uncertainty in position is twice the uncertainty in momentum. Uncertainty in velocity is :
Options
A.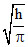
B.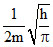
C.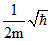
D.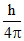
Solution
ᐃx = 2ᐃp
ᐃx . ᐃp =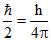 ⇒ 2ᐃp . ᐃp =
⇒ 2ᐃp . ᐃp = 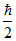
⇒ 2(mᐃV)2 =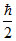 ; (ᐃV)2 =
; (ᐃV)2 = 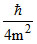 ⇒ ᐃV =
⇒ ᐃV = 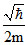 .
.
ᐃx . ᐃp =
⇒ 2(mᐃV)2 =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Among the following, the correct statement(s) is(are)...The e/m ratio of anode rays produced in the discharge tube, depends on the...An α-particle accelerated through V volt is fired towards a nucleus. The distance of closest approach is r. If a proton ...As its closest approach, the distance between the Mars and the Earth is found to be 60 million km. When the planets are ...Which of the following ion is diamagnetic?...