Atomic StructureHard
Question
The total spin resulting from a d7 configuration is :
Options
A.1
B.2
C.5/2
D.3/2
Solution
d7 : 3 unpaired electrons.
∴ Total spin = ±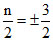
∴ Total spin = ±
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Simultaneous determination of exact position and momentum of an electron is :-...In H-atom, if ′x′ is the radius of the first Bohr orbit, de Broglie wavelength of an electron in 3rd orbit i...A photon of energy hn is absorbed by a free electron of a metal having work function w < hn. Then :...If ᐃ0 < P.E. the correct electronic configuration for d6 system will be :-...If the value of En = - 78.4 kcal/mole, the order of the orbit in hydrogen atom is :...