Atomic StructureHard
Question
The orbital with zero orbital angular momentum is-
Options
A.s
B.p
C.d
D.f
Solution
Orbital angular momentum = 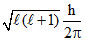 = 0 ∴ l = 0 (s orbital).
= 0 ∴ l = 0 (s orbital).
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The ratio of spacing between the third and fourth orbit to the spacing between sixth and seventh orbit of H–atom is...The magnetic moment of Nix+ ion (Z = 28) is about 2.82 BM. The value of x is...Which of the following statements is/are correct ?1.0 g mixture of CaCO3(s) and glass beads liberate 0.22 g of CO2 upon ...The energy of hydrogen atom in its ground state is -13.6 eV. The energy of the level corresponding to n = 5 is :...The size of an orbital is given by...