Atomic StructureHard
Question
Which of the following statements is/are correct ?
1.0 g mixture of CaCO3(s) and glass beads liberate 0.22 g of CO2 upon treatment with excess of HCl. Glass does not react with HCl.
CaCO3 + 2HCl → CO2 + H2O + CaCl2
[Mw CaCO3 = 100, Mw of CO2 = 44, [Atomic weight of Ca = 40]
1.0 g mixture of CaCO3(s) and glass beads liberate 0.22 g of CO2 upon treatment with excess of HCl. Glass does not react with HCl.
CaCO3 + 2HCl → CO2 + H2O + CaCl2
[Mw CaCO3 = 100, Mw of CO2 = 44, [Atomic weight of Ca = 40]
Options
A.The weight of CaCO3 in the original mixture is 0.5 g.
B.The weight of calcium in the original mixture is 0.2 g,
C.The weight percent of calcium in the original mixture is 40% Ca.
D.The weight percent of Ca in the original mixture is 20% Ca.
Solution
a. Weight of CaCO3 = (0.22g CO2)
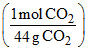
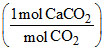

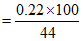 = 0.5 g CaCO3
= 0.5 g CaCO3
b. Moles of CaCO3 = moles of Ca
=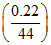 = 0.005 mol
= 0.005 mol
weight Ca = 0.005 × 40 = 0.2 g Ca
d. % of Ca =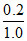 × 100 = 20% Ca
× 100 = 20% Ca
Hence, (c) is wrong.
b. Moles of CaCO3 = moles of Ca
=
weight Ca = 0.005 × 40 = 0.2 g Ca
d. % of Ca =
Hence, (c) is wrong.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The number of nodal planes in a Px orbotal is :...For an electron, with n = 3 has only one radial node. The orbital angular momentum of the electron will be...The energy of first (lowest) Balmer line of H atom is x J . The energy (in J) of second Balmer line of H atom is :...Wavelength of photon having energy 1 eV would be...No. of visible lines when an electron returns from 5th orbit to ground state in H spectrum -...