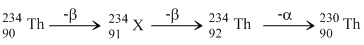Atomic StructureHard
Question
The radionucleide 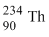 undergoes two successive β - decays followed by one α - decay. The atomic numberand the mass number respectively of the resulting radionucleide are
undergoes two successive β - decays followed by one α - decay. The atomic numberand the mass number respectively of the resulting radionucleide are
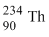 undergoes two successive β - decays followed by one α - decay. The atomic numberand the mass number respectively of the resulting radionucleide are
undergoes two successive β - decays followed by one α - decay. The atomic numberand the mass number respectively of the resulting radionucleide areOptions
A.94 and 230
B.90 and 230
C.92 and 230
D.92 and 234
More Atomic Structure Questions
Maximum number of electrons in a subshell with l = 3 and n = 4 is...In which of the following process e- is removed form σ2p orbital...de-Broglie wavelength of electron in second orbit of Li2+ ion will be equal to de-Broglie of wavelength of electron in...The respective values of orbital angular momentum of an electron in 1s, 2s, 3d and 3p-orbitals are:-...Photon of which light has maximum energy :...