Atomic StructureHard
Question
What is likely to be orbit number for a circular orbit of diameter 20 nm of the hydrogen atom if we assume Bohr orbit to be the same as that represented by the principal quantum number ?
Options
A.10
B.14
C.12
D.16
Solution
Radius = 0.529 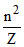 (Ao) = 10 × 10-9 m
(Ao) = 10 × 10-9 m
So, n2 = 189 or, n ≈ 14
So, n2 = 189 or, n ≈ 14
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
A lithium atom has three electrons. Assume the following simple picture of the atom. Two electrons move close to the nuc...In which transition, one quantum of energy is emitted ?...Photon having wavelength 310 nm is used to break the bond of A2 molecule having bond energy 288 kJ mol-1 then % of energ...The correct order of atomic radii in group 13 elements is...The ratio of wavelengths of Kα-characteristic X-rays produced when iron (Z = 26) and scandium (Z = 21) are used as antic...