ElectrostaticsHard
Question
Consider the following reaction:
x MnO4- + y C2O42- + z H+ → x Mn2+ + (2y) CO2 + (z/2) H2O
The values of x, y and z in the reaction are respectively :-
Options
A.5,2 and 16
B.2,5 and 8
C.2, 5 and 16
D.5,2 and 8
Solution
(i) MnO4- + 8H+ + 5e- → Mn+2 + 4H2O
(ii) C2O4-2 → 2CO2 + 2e-
⇒ (i)2 + (ii) 5

2MnO4- + 16H+ + 5C2O4-2 → 2Mn+2 + 8H2O + 10CO2

Given equation :
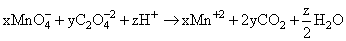
On comparing with balanced equation
we get ; x = 2, y = 5, z = 16.
Create a free account to view solution
View Solution FreeMore Electrostatics Questions
How many times, the potential of big drop in comparison to small drops which is made of 8 droplets will be, if all the d...The electric field produced by a positively charged particle, placed in an xy-plane is 7.2 (4î + 3ĵ) N/C at th...A insulating cylindrical rod of diameter d and length l(l >> d) has a uniform surface charge density such that the elect...A charged particle of charge ′Q′ is held fixed and another charged particle of mass ′m′ and char...Four electric charge +q, +q, -q and -q are placed at the corner of a square of side 2 L (see figure). The electric poten...